| |
| Med Sci (Paris). 34: 59–65. doi: 10.1051/medsci/201834f111.miR-25 Promotes Melanoma Progression by regulating RNA binding motif protein 47 Qun-Qun Jiang1* and Wei-Bing Liu1 1Department of Dermatology, 404 Hospital of People’s Liberation Army, No.8 of Baoquan Street, Huancui District, Weihai, 264200, Shandong Province, China |
Melanoma is the most aggressive skin cancer, and accounts for the major part of skin cancer-related deaths in the world. In addition, the underlying mechanism of tumor progression in melanoma remains far from being elucidated. In this study, we have evaluated the function of miR-25 in melanoma. First, we examined the expression of miR-25 in four melanoma cell lines (A875, MV3, M14 and uacc-257) and in a normal melanocyte cell line (HEM-a). Then, we overexpressed miR-25 in M14 cells. Our results show that miR-25 promotes M14 cell proliferation and migration. We found that miR-25 up-regulates the PI3K/Akt/mTOR signaling pathway in these tumor cells. Furthermore, a luciferase-based reporter gene assay showed that miR-25 could directly target the RNA-binding motif protein 47 (RBM47). Taken together, our findings suggest that RBM47 is a promising target for the treatment of melanoma. Keywords: melanoma cell, migration, miR-25, RBM47, proliferation |
Skin cancer is one of the most common cancer in the world. Basal cell carcinoma, squamous cell carcinoma and melanoma are all skin cancers [1-3]. Melanoma rarely occurs and only accounts for 2.3% of all skin cancers, but it is the most dangerous. 75% of skin cancer-related deaths are caused by melanoma [4]. Melanomas are usually related to ultraviolet radiation and arise in skin. Superficial spreading, acral lentiginous, nodular and lentigomaligna melanomas are four clinical categories [5]. Despite major surgery can remove melanoma successfully, the identity of primary melanoma cells invading surrounding tissues and the treatment efficacy for metastatic melanoma are poor [6, 7]. In addition, cancer cells always develop drug resistance to clinical treatments. Hence, there is an urgent need to find novel strategies to ameliorate melanoma treatment. Many studies have shown that miRNAs regulate various processes in tumorigenesis, including cell proliferation, migration and invasion. miR-25 has been found significantly overexpressed in various tumors [8, 9]. Previous studies have demonstrated that miR-25 promotes cancer cell proliferation, migration and invasion by regulating the expression of various protein in tumor-associated signaling pathways [10-13]. However, another study demonstrated that miR-25 suppresses cell growth and mobility of osteosarcoma via targeting SOX4 [14]. In addition, other studies have shown that miR-25 inhibits cell apoptosis in various cancers, including gastric adenocarcinoma, lung cancer and pancreatic cancer [15-18]. Thus, despite many reports have demonstrated the effects of miR-25 in tumorigenesis, the involvement and impact of miR-25 in the progression of melanoma remain unexplored in details. RNA binding motif protein 47 (RBM47) is critical for posttranscriptional regulation of RNA during embryonic development and tumor progression. Recent studies have shown that RBM47 exerts diverse effects in embryonic development. It has been reported that knockdown of RBM47 gene causes headless phenotype during zebrafish head development [19]. In addition, RBM47 contributes to somatic cell reprogramming through the control of alternative splicing (AS) or binding to transcription factorss [20, 21]. RBM47 is considered to be an epithelial cell state-associated gene and its down-regulation increases the metastatic traits of colorectal, breast and lung cancer [22-24]. RBM47 also inhibits cancer cell growth through the targeting of Nrf2 in lung adenocarcinoma [25]. So far, the relationship between RBM47 gene and melanoma development is unclear. In the present study, we have investigated the function of miR-25 in melanoma by targeting RBM47. First, we evaluated the expression of miR-25 in melanoma cell lines (A875, MV3, M14 and uacc257). Then we overexpressed miR-25 in M14 cells. Our results show that miR-25 promotes M14 cell proliferation and migration. It also up-regulates the PI3K/AKT/mTOR signaling pathway in melanoma. We also demonstrate herein that RBM47 is a direct target of miR-25. All of these findings demonstrated that RBM47 might be a new drug target for the treatment of melanoma. |
Cell culture Human melanoma cell lines, M14, A875, MV3, uacc257 and the normal melanocyte HEM-a were purchased from the Type Culture Collection of the Chinese Academy of Sciences, Shanghai, China. All cells were cultured in RPMI 1640 medium (Hyclone) containing 10% fetal bovine serum (Gibco), 0.1 mg/mL streptomycin and 100 U/mL penicillin (Sigma). Cells were cultured in a 5% CO 2 incubator at 37 °C. Cells were used for further experiment while in their exponential growth phase. Cells were washed with PBS for 3 times and then treated with trypsin (Solarbio), after that resuspended cells were seeded into 6-well plates. The transfection was performed when cell density reached 80%. Transfection DNA transfection was performed by using Lipofectamine 2000 Transfection Reagent (Invitrogen) following the manufacturer’s instructions. Cells on 6-well plate were changed with fresh completed medium 2 hours before transfection. 10 μL Lipofectamine 2000 reagents were diluted in 250 μL serum-free medium, mixed well gently and incubated for 5 min at room temperature. 2.5 μg pCMV-MIR-miR25 plasmid and control vector pCMV-MIR plasmid (Negative control, NC) were diluted in 250 μL serum-free medium. Then diluted DNA was added into Lipofectamine 2000 reagent, mixed well and incubated for 30 minutes at room temperature. The medium of 6-well plates were discarded, and the cells were washed with PBS for 3 times. Then about 500 μL mixture solutions were added into 6-well plates. After 6 hours culture, the cells were changed to the complete medium. The following experiments were performed after 24 hours culture. RNA extraction and quantitative real-time PCR assay Total RNA was extracted by using Trizol reagent (CWBIO) following the manufacturer’s instructions. 1 microgram of total RNA was used for cDNA synthesis. miR-25 expression was assessed by quantitative real-time PCR with the SYBR Green PCR Master Mix (CWBIO) according to the manufacturer’s protocol. The qRT-PCR reactions were performed at 95 °C for 5 min, followed by 40 cycles of 95 °C for 30 sec and 60 °C for 45 sec, and, finally, maintained at 72 °C for 30 min. The U6 was used to normalize expression: U6 forward, 5’-CTCGCTTCG GCAGCACA-3’ and U6 reverse, 5’-AACGCTTCACGAATTTGCGT-3’. miR-25 primers were purchased from Guangzhou RiboBio Co. All reactions were performed in triplicate. Data analysis was performed according to the 2 -ΔΔCt method. Western blot M14 melanoma cells were harvested 48 hours after transfection. Cells were washed with cold PBS and lysed in RIPA buffer (CWBIO) with protease inhibitors (CWBIO). Protein concentration was measured by BCA protein determination method (CWBIO). Protein samples (20 μg) were loaded on SDS-PAGE gel and transferred onto PVDF membrane (Millipore). After transfer, the PVDF membrane was blocked with 5% non-fat dried milk for 1 h, and then incubated with primary antibodies overnight (4°C) and then with HRP-conjugated secondary antibody for 1 hour at room temperature. Finally, the protein bands were visualized by ECL reagents (Pierce). GAPDH was used as a house-keeping gene. Reference antibodies RBM47 (Abcam, ab167164), AKT (Proteintech, 60203-2-Ig), p-AKT (Proteintech, 66444-1-lg), mTOR (Affinity, AF6308), p-mTOR (Affinity, AF3308), P70 (Proteintech, 14485-1-AP), Cyclin D1 (Proteintech, 60186-1-lg), and GAPDH (Proteintech, 60004-1-lg) were used. Data were analyzed with the Quantity One software. Cell Counting Kit-8 assay M14 cells were cultured for 24 hours after transfection. The cells were then treated with trypsin, resuspended and counted. 1,000 cells were seeded into 96-well plates in 100 μL medium and cultured in a 5% CO 2 incubator at 37 °C. Cell viability was tested every 24 hours. For each test, 10 μL CCK-8 (Solarbio) reagent was added into the wells and incubated for 1.5 hour at 37 °C. Finally, the absorbance (optical density, O.D.) was measured with a microplate reader at 450 nm. Plate clone formation assay M14 cells were resuspended and counted 24 hours after transfection. They were then seeded into 60 mm plates (500 cells/plate) in 5 mL pre-warmed medium. The plates were incubated for 2 weeks in a 5% CO 2 incubator at 37 °C. During this period, complete medium (1 mL) was added into each well of the plate every 2~3 days. Cells were then fixed with 4% paraformaldehyde for 30 minutes and stained with 0.1% crystal violet for another 30 minutes. Cell clones were counted and analyzed after washing. Transwell migration assay M14 cells were resuspended in serum-free medium 24 hours after transfection. For the migration assay, 10 5 cells in 100 µL serum-free medium were seeded into the upper chamber. 500 µL complete medium was added to the bottom wells to stimulate migration or invasion. After incubation, the non-invasive cells in the upper chamber were removed with cotton swabs. The invading cells were fixed with 4% paraformaldehyde for 30 minutes and stained with 0.1% crystal violet. Five fields per filter were counted using microscope. All samples were performed in triplicate. Dual-luciferase reporter assay The wild-type ( wt) or mutated RBM47 3’UTR were inserted into pmirGLO vector. The pmirGLO-RBM47 3’UTR ( wt) or pmirGLO-RBM47 3’UTR (mut) vectors were co-transfected with pCMV-MIR-miR25 or with a negative control (pCMV-MIR) into M14 cells by lipofectamine 2000. Forty-eight hours after transfection, the luciferase activity was measured using the Dual-Luciferase Reporter Assay System (Promega). Statistical analysis Statistical analysis was carried out using the SPSS 18.0 software. Data were presented as mean ± SD. The Student’s t test (2-tailed) or one-way ANOVA were used to determine the significance of different groups. P<0.05 was considered statistically significant. |
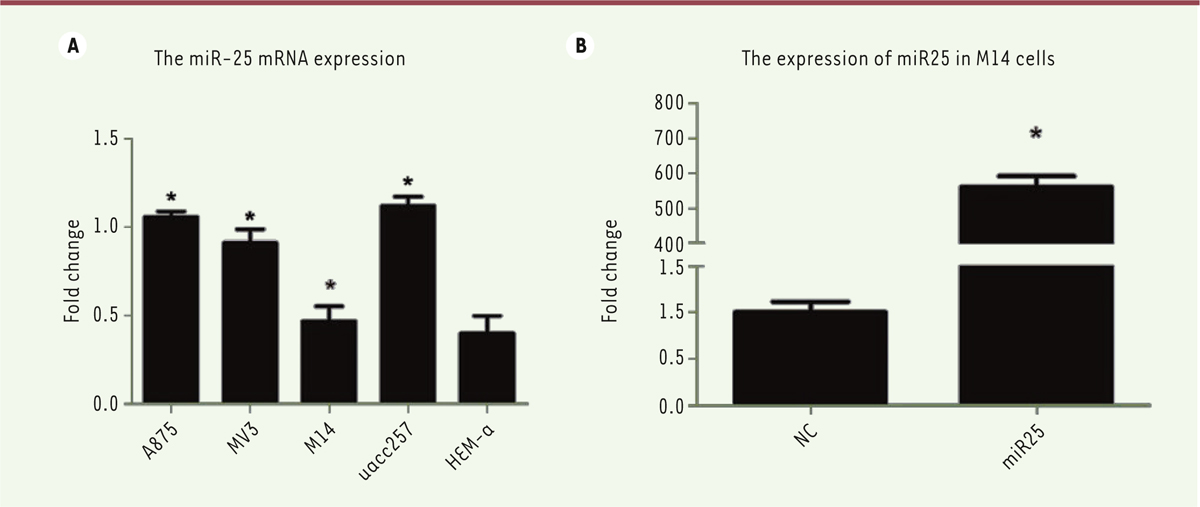
Expression levels of miR-25 in cells from various melanoma cell lines So far, only a few reports have been published on the functions of miR-25. It contributes to the decrease of cardiac function during heart failure [ 26], regulates intracellular calcium homeostasis, protects from Ca 2+-dependent apoptosis [ 27], targets TNF-related apoptosis-inducing ligand (TRAIL) that induces death receptor-4 network and promotes apoptosis resistance [ 28]. Moreover, miR-25 inhibits apoptosis in human cholangiocarcinoma [ 28] and ovarian cancer [ 9]. In the present study, we tried to research the function and mechanism of miR-25 in melanoma cells. First, we selected 4 melanoma cell lines and a normal melanocyte line as control to detect the miR-25 transcription level. Using quantitative real-time PCR assay, we found that miR-25 widely exists in normal melanocytes and cells from melanoma cell lines. In addition, the expression level of miR-25 in cells from all melanoma cell lines was stronger than in normal melanocyte, HEM-a (P<0.05). However, the miR-25 level was lower in M14 cells than in the cells from the three other melanoma cell lines, A875, MV3 and uacc257 (Figure 1A). Because of this lower expression of miR-25, we selected the M14 cell line to overexpress miR-25 to further investigate its functions in melanoma. As shown in Figure 1B, the expression level of miR-25 was dramatically increased after exogenous plasmid transfection compared with control (non-transfected M14 cells).
 | Figure 1. Expression level of miR-25 in melanoma cell lines. (A) miR-25 endogenous expression in A875, MV3, M14 and UACC-257 melanoma cells and HEM-a normal melanocytes were determined by real-time PCR. (B) Overexpression of miR-25 in M14 cells following transfection. Results represent data from three independent experiments. * P<0.05. P values were determined using Student’s t-tests. |
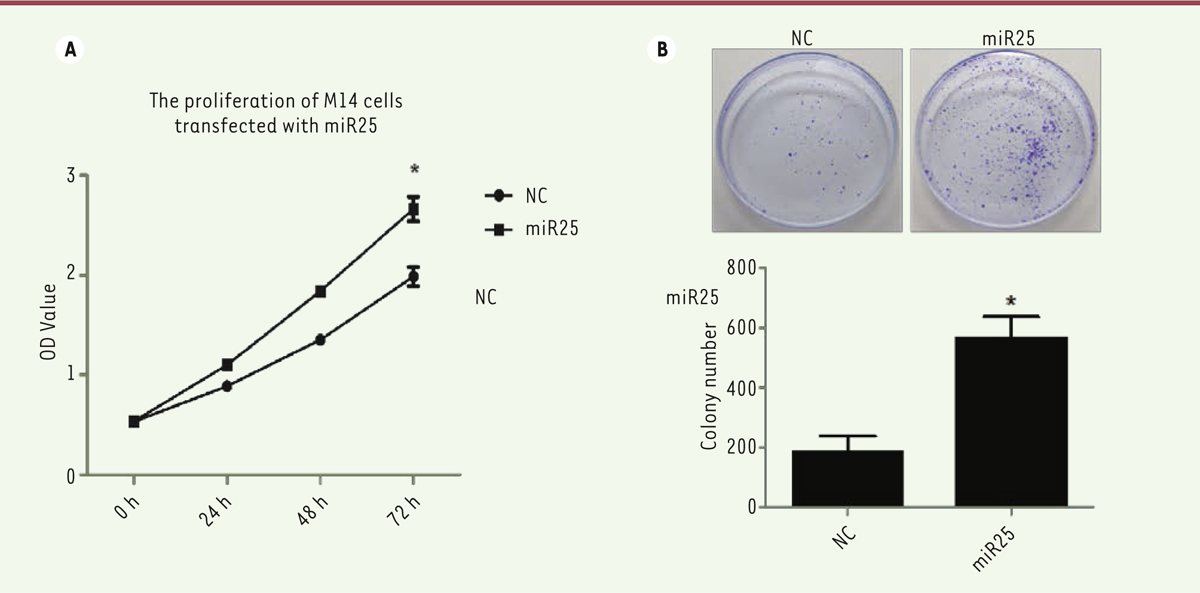
miR-25 overexpression increases melanoma M14 cell proliferation To determine the effect of miR-25 on cell proliferation, we counted M14 cells using the kit-8 assay (CCK8) and plate clone formation assay after miR-25 or NC plasmid transfection. The CCK8 assay showed that the proliferation of M14 cells transfected with miR-25 increased after 72 hours transfection compared to cells transfected with the control vector (P<0.05, Figure 2A). Similarly, the plate clone formation assay showed that the M14 cell clone number is significantly increased when miR-25 is overexpressed as compared with cells transfected with the control vector (P<0.05, Figure 2B). Thus, the overexpression of miR-25 promotes M14 cell proliferation.
 | Figure 2. A strong expression of miR-25 is associated with an increase of melanoma M14 cell proliferation. (A) CKK-8 assay of M14 cells proliferation after miR-25 plasmid or NC plasmid transfection. (B) Colony formation assay of M14 cells transfected with NC or miR-25 plasmids. Cells were fixed and stained with Giemsa. One representative experiment out of three is shown. * P<0.05. P values were determined using Student’s t-tests. |
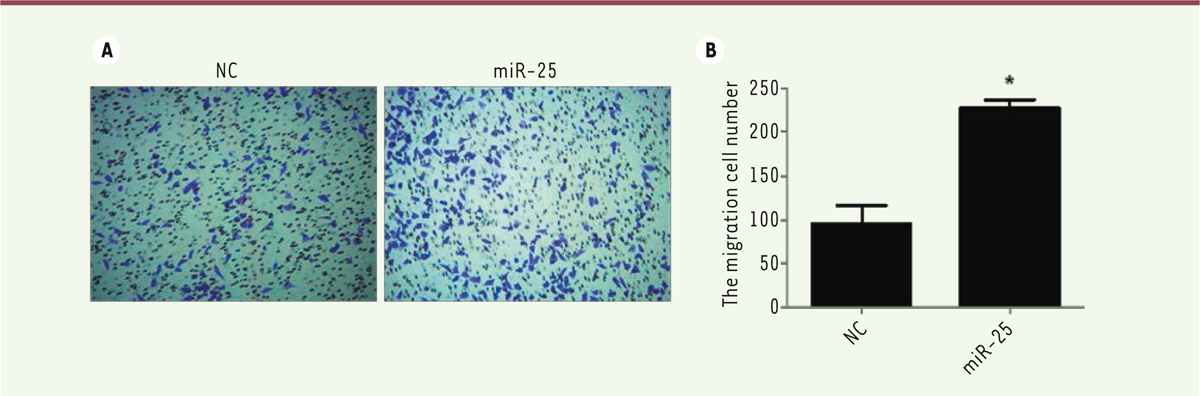
A strong expression of miR-25 is associated with an increase in melanoma M14 cell migration A common feature of cancer cells is their aberrant motility, leading to cell migration. To further investigate the effect of miR-25 in regulating cell motility, we performed Transwell assays to evaluate the role of miR-25 on M14 cell migration. As shown in Figure 3A and B, the migration ability of miR-25 transfected M14 cells was significantly increased as compared to the negative control group (P<0.05). These results suggest that miR-25 is able to increase the ability of melanoma cell migration.
 | Figure 3. A strong expression of miR-25 increases melanoma M14 cell migration. (A) M14 cells were transfected with NC or miR-25 plasmids. Cell migration was analyzed by a transwell assay where a 10% FBS culture medium was used as chemoattractant. (B) Statistical analysis of cell numbers after 0.1% crystal violet staining. Results represent data from three independent experiments. * P<0.05. P values were determined using Student’s t-tests. |
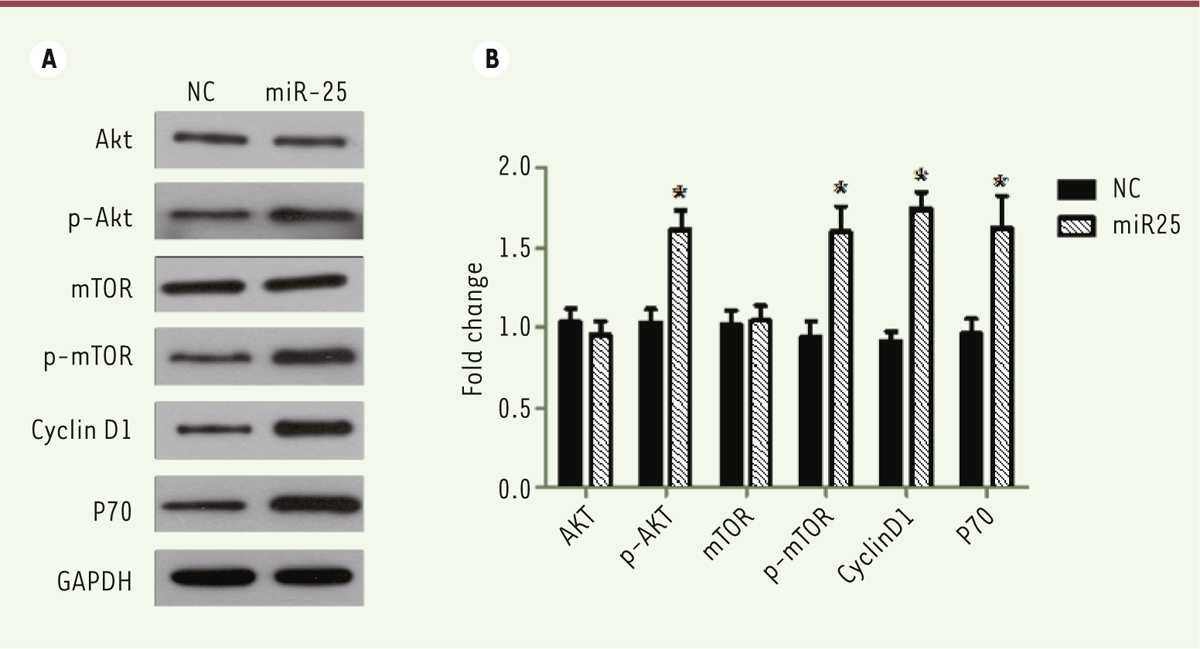
miR-25 up-regulates the PI3K/Akt/mTOR signaling pathway in melanoma M14 cells We then investigated which signaling pathway could be involved in the effects on proliferation and migration observed when miR-25 expression is increased in M14 melanoma cells. The PI3K/Akt/mTOR signaling pathway is an important regulator of physiological cell processes which include proliferation, motility, differentiation, metabolisms and cell death. Figure 4 shows that the activation of PI3K/Akt/mTOR signaling pathway is associated with an increased expression and function of miR-25 in M14 cells. Western blots indicated that the amount of phosphorylated AKT (p-Akt), phosphorylated mTOR (p-mTOR), Cyclin D1 and P7 are increased (P<0.05, Figure 4A and B), as compared to cells transfected with the control vector. Altogether, these data suggest that PI3K/Akt/mTOR signaling pathway is involved in the effects of miR-25 on melanoma cell proliferation and cell motility.
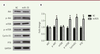 | Figure 4. miR-25 up-regulates the PI3K/AKT/mTOR signaling pathway in melanoma M14 cell. (A) M14 cells were transfected with NC or miR-25 plasmids for 24 hours and expression of AKT, p-AKT, mTOR, p-mTOR, Cyclin D1, P70 and GAPDH were detected by western blots. (B) The relative amounts of proteins were normalized using GAPDH. Results represent data from three independent experiments. * P<0.05. P values were determined using Student’s t-tests. |
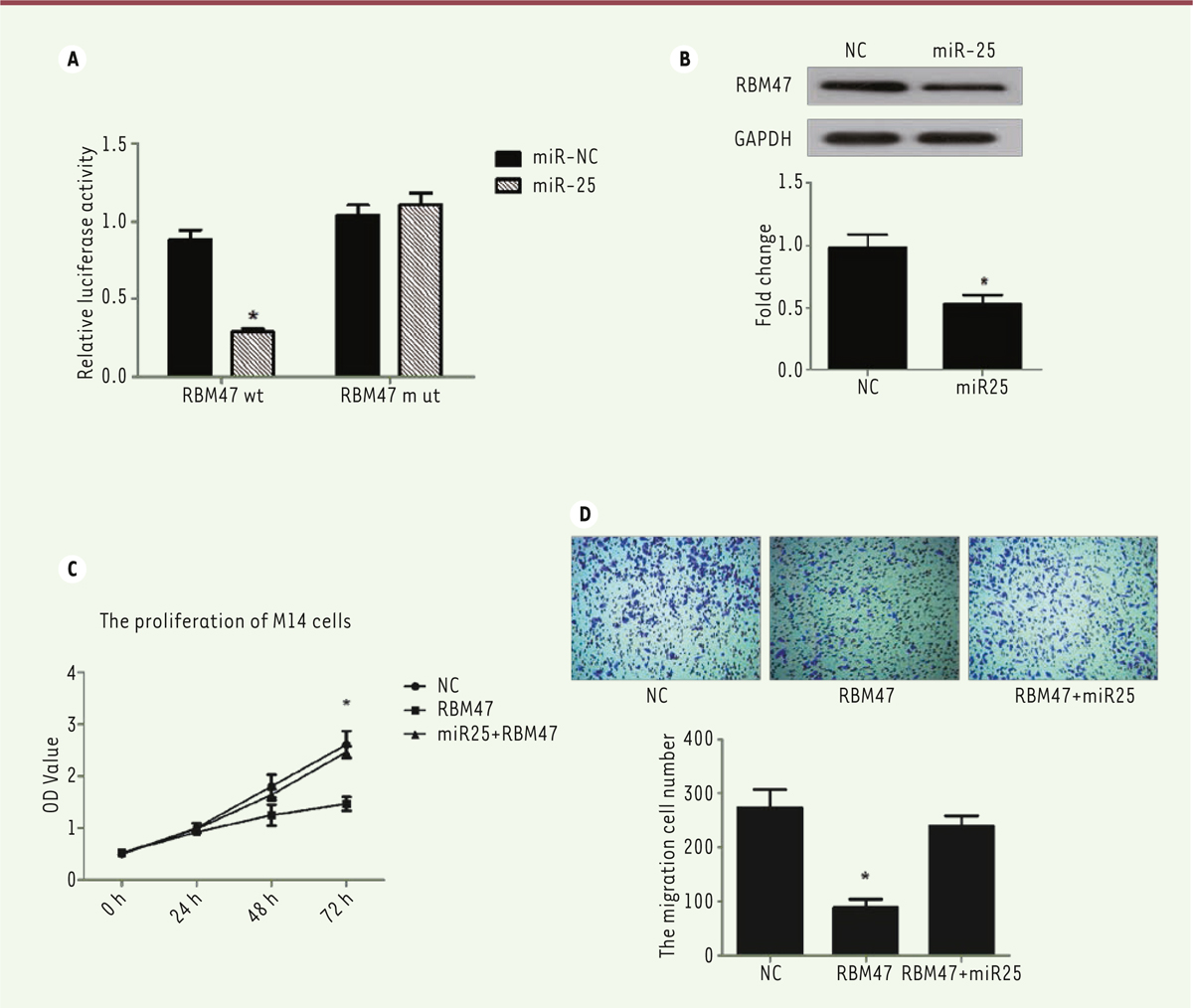
miR-25 targets RNA binding motif protein 47 Matured miRNAs are single-stranded RNAs and act through a perfect or near perfect base pairing with the target mRNA 3'-untranslated regions (3'-UTRs), leading to translational repression and/or mRNA cleavage. Thus, we investigated which sequence miR-25 targets in melanoma cells. We found that RNA-binding protein 47 (RBM47) could be a direct target of miR-25 through an online analysis using Targetscan ( http://www.targetscan.org/vert_72/), This tool makes it possible to predict biological targets of miRNAs by searching for the presence of conserved sites that match the seed region of miRNAs. In order to verify our hypothesis, we used a luciferase-based reporter gene assay to test the relationship of miR-25 and RBM47. Our data showed that miR-25 could directly target the RBM47 promoter and inhibit its transcription function. In addition, miR-25 had no effect on the mutant RBM47 promoter (P< 0.05, Figure 5A). Meanwhile, Western blot experiment showed that miR-25 decreases RBM47 protein expressionl(P <0.05, Figure 5B). Thus, RBM47 appears to be a direct target of miR-25.
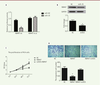 | Figure 5. miR-25 targets the RNA-binding protein 47. (A) Luciferase-based reporter gene assay detects RBM47 inhibition by miR-25 through the targeting of its promoter. (B) miR-25 inhibits RBM47 protein expression in M14 cells. (C) CKK-8 assay of M14 cell proliferation after co-transfection of miR-25 plasmid or NC plasmid with RBM47 expression plasmid or empty plasmid used as a control. (D) Transwell migration assay of M14 cells after co-transfection of miR-25 plasmid or NC plasmid with RBM47 expression plasmid or empty plasmid used as a control. 10% FBS was used as the chemoattractant. *P < 0.05. P values were determined using Student’s t-tests. |
To further confirm that RBM47 is a target of miR-25, we transfected a RBM47-encoding plasmid or together with a miR-25-encoding plasmid (or an empty plasmid as a control). in M14 melanoma cells. Strikingly, miR-25 could rescue RBM47-induced suppression of cell proliferation (P<0.05, Figure 5C) and migration (P<0.05, Figure 5D). Thus, miR-25 promoted melanoma cell M14 proliferation and motility by directly targeting and inhibiting RBM47. |
Melanoma is the most aggressive skin cancers and is the first leading cause of skin cancer-related death in the world [4]. Advances in diagnosis and clinical therapy have enable to reach a 5-year survival of melanoma patients, but the therapy of metastatic melanoma is extremely limited [5]. In addition, melanoma progression still remains poorly understood. In order to find more effective therapeutic strategy for treating melanoma cancer, it is important to identify novel melanoma-related factors which are responsible for cancer incidence and progression and to unravel the underlying mechanisms. Hence, there is an urgent need to find novel factors associated with melanoma and strategies to improve melanoma treatment. Previous studies have shown that miR-25 plays a critical role in tumorigenesis and in gastric carcinogenesis. The expression of miR-25 in many gastric cancer patient tissues and plasma is significantly increased [29, 30]. Interestingly, cyclin-dependent kinase 6 (CDK6) plays a key role in mammalian cell proliferatio and previous studies indicate that miR-25 regulates CDK6 directly in vascular smooth muscle cell, hence, inhibiting cell proliferation and ki-67 expression [31]. In addition, another cell cycle-related protein p27 is regulated by miR-25 negatively, and was shown to promote proliferation of osteosarcoma cells [32]. Abnormal expression of miR-25 in several types of cancer suggests its potential association with tumorigenesis. To date, the association between miR-25 and melanoma remains unclear. RNA binding motif protein 47 (RBM47) is critical for epithelial mesenchymal transition during embryonic development and tumor progression. RBM47 together with its cofactor catalytic polypeptide-1 (APOBEC1) are required for cellular processes such as the transendothelial migration and the C to U RNA editing [33-36]. Our study indicates for the first time that miR-25 directly targeted to RBM47. The A875, MV3, M14 and uacc257 melanoma cancer cells and the normal melanocytes HEM-a were selected to perform the present study. We first evaluated the expression level of miR-25 in the cells from the five cell lines (Figure 1). Our results showed that miR-25 expression is up-regulated in melanoma cancer cells compared with normal melanocytes. Since the expression level of miR-25 in M14 cells was the lowest among melanoma cell lines, we overexpressed miR-25 in these cells to further investigate miR-25 functions (Figure 1). We found that M14 cell proliferation is increased in cells transfected with a miR-25-encoding expression vector (Figure 2). Similarly, results of plate clone formation assay showed that the cell clone number was significantly increased with miR-25 overexpression (Figure 2). Western blots showed that a high amount of miR-25 is associated with the activation of the PI3K/Akt signaling pathway marked by an increase of the expression of phosphorylated Akt (p-Akt), phosphorylated mTOR (p-mTOR), Cyclin D1 and P70 (Figure 4). Finally, we showed that a high level of miR-25 is associated with an increase in melanoma cell M14 proliferation and motility by directly targeting and inhibiting RBM47 (Figure 5). In conclusion, our study demonstrates that a strong expression of miR-25 in M14 cells is associated with an increased proliferation and migration by directly targeting and inhibiting RBM47. Inhibition of miR-25 may be therefore a candidate approach for treatment of melanoma and new drug targeting RBM47 is worthy of further study and exploration. |
The authors declare that they have no conflict of interest. |
1. Perna FM, Dwyer LA, Tesauro G, et al. Research on Skin Cancer-Related Behaviors and Outcomes in the NIH Grant Portfolio, 2000–2014: Skin Cancer Intervention Across the Cancer Control Continuum (SCI-3C) . JAMA Dermatol. 2017; ; 153 : :398.–405. 2. The Surgeon General‘s Call to Action to Prevent Skin Cancer . Washington (DC): 2014. 3. Luke JJ, Triozzi PL, McKenna KC, et al. Biology of advanced uveal melanoma and next steps for clinical therapeutics . Pigment Cell Melanoma Res. 2015; ; 28 : :135.–147. 4. Corrie P, Hategan M, Fife K, Parkinson C. Management of melanoma . Br Med Bull. 2014; ; 111 : :149.–162. 5. Situm M, Buljan M, Kolic M, Vucic M. Melanoma–clinical, dermatoscopical, and histopathological morphological characteristics . Acta Dermatovenerol Croat. 2014; ; 22 : :1.–12. 6. Jaimes N, Marghoob AA. The morphologic universe of melanoma . Dermatol Clin. 2013;; 31 : :599.–613, viii-ix.. 7. Dao AH, Page DL, Reynolds VH, Adkins RB, Jr.. Primary malignant melanoma of the adrenal gland. A report of two cases and review of the literature . Am Surg. 1990; ; 56 : :199.–203. 8. Marchi S, Pinton P. Mitochondrial calcium uniporter, MiRNA and cancer: Live and let die . Commun Integr Biol. 2013; ; 6 : :e23818.. 9. Zhang H, Zuo Z, Lu X, et al. MiR-25 regulates apoptosis by targeting Bim in human ovarian cancer . Oncol Rep. 2012; ; 27 : :594.–598. 10. Gong J, Cui Z, Li L, et al. MicroRNA-25 promotes gastric cancer proliferation, invasion, and migration by directly targeting F-box and WD-40 Domain Protein 7, FBXW7 . Tumour Biol. 2015; ; 36 : :7831.–7840. 11. Wang C, Wang X, Su Z, et al. MiR-25 promotes hepatocellular carcinoma cell growth, migration and invasion by inhibiting RhoGDI1 . Oncotarget. 2015; ; 6 : :36231.–36244. 12. Xiang J, Hang JB, Che JM, Li HC. MiR-25 is up-regulated in non-small cell lung cancer and promotes cell proliferation and motility by targeting FBXW7 . Int J Clin Exp Pathol. 2015; ; 8 : :9147.–9153. 13. Huo J, Zhang Y, Li R, et al. Upregulated MicroRNA-25 Mediates the Migration of Melanoma Cells by Targeting DKK3 through the WNT/beta-Catenin Pathway . Int J Mol Sci. 2016; ; 17 : 14. Wu X, Zhou H, Yue B, et al. Upregulation of microRNA-25-3p inhibits proliferation, migration and invasion of osteosarcoma cells in vitro by directly targeting SOX4 . Mol Med Rep. 2017; ; 16 : :4293.–4300. 15. Feng X, Jiang J, Shi S, et al. Knockdown of miR-25 increases the sensitivity of liver cancer stem cells to TRAIL-induced apoptosis via PTEN/PI3K/Akt/Bad signaling pathway . Int J Oncol. 2016; ; 49 : :2600.–2610. 16. Zhang Y, Peng Z, Zhao Y, Chen L. microRNA-25 Inhibits Cell Apoptosis of Human Gastric Adenocarcinoma Cell Line AGS via Regulating CCNE1 and MYC . Med Sci Monit. 2016; ; 22 : :1415.–1420. 17. Zhang JF, Shi LL, Zhang L, et al. MicroRNA-25 Negatively Regulates Cerebral Ischemia/Reperfusion Injury-Induced Cell Apoptosis Through Fas/FasL Pathway . J Mol Neurosci. 2016; ; 58 : :507.–516. 18. Chen Z, Wu Y, Meng Q, Xia Z. Elevated microRNA-25 inhibits cell apoptosis in lung cancer by targeting RGS3 . In Vitro Cell Dev Biol Anim. 2016; ; 52 : :62.–67. 19. Guan R, El-Rass S, Spillane D, et al. rbm47, a novel RNA binding protein, regulates zebrafish head development . Dev Dyn. 2013; ; 242 : :1395.–1404. 20. Yeganeh M, Seyedjafari E, Kamrani FA, Ghaemi N. RNA-binding protein Rbm47 binds to Nanog in mouse embryonic stem cells . Mol Biol Rep. 2013; ; 40 : :4391.–4396. 21. Cieply B, Park JW, Nakauka-Ddamba A, et al. Multiphasic and Dynamic Changes in Alternative Splicing during Induction of Pluripotency Are Coordinated by Numerous RNA-Binding Proteins . Cell Rep. 2016; ; 15 : :247.–255. 22. Rokavec M, Kaller M, Horst D, Hermeking H. Pan-cancer EMT-signature identifies RBM47 down-regulation during colorectal cancer progression . Sci Rep. 2017; ; 7 : :4687.. 23. Vanharanta S, Marney CB, Shu W, et al. Loss of the multifunctional RNA-binding protein RBM47 as a source of selectable metastatic traits in breast cancer . Elife. 2014;; 3. 24. Yang Y, Park JW, Bebee TW, et al. Determination of a Comprehensive Alternative Splicing Regulatory Network and Combinatorial Regulation by Key Factors during the Epithelial-to-Mesenchymal Transition . Mol Cell Biol. 2016; ; 36 : :1704.–1719. 25. Sakurai T, Isogaya K, Sakai S, et al. RNA-binding motif protein 47 inhibits Nrf2 activity to suppress tumor growth in lung adenocarcinoma . Oncogene. 2016; ; 35 : :5000.–5009. 26. Wahlquist C, Jeong D, Rojas-Munoz A, et al. Inhibition of miR-25 improves cardiac contractility in the failing heart . Nature. 2014; ; 508 : :531.–535. 27. Marchi S, Lupini L, Patergnani S, et al. Downregulation of the mitochondrial calcium uniporter by cancer-related miR-25 . Curr Biol. 2013; ; 23 : :58.–63. 28. Razumilava N, Bronk SF, Smoot RL, et al. miR-25 targets TNF-related apoptosis inducing ligand (TRAIL) death receptor-4 and promotes apoptosis resistance in cholangiocarcinoma . Hepatology. 2012; ; 55 : :465.–475. 29. Espinosa-Parrilla Y, Munoz X, Bonet C, et al. Genetic association of gastric cancer with miRNA clusters including the cancer-related genes MIR29, MIR25, MIR93 and MIR106: results from the EPIC-EURGAST study . Int J Cancer. 2014; ; 135 : :2065.–2076. 30. Zhang R, Wang W, Li F, et al. MicroRNA-106b~25 expressions in tumor tissues and plasma of patients with gastric cancers . Med Oncol. 2014; ; 31 : :243.. 31. Qi L, Zhi J, Zhang T, et al. Inhibition of microRNA-25 by tumor necrosis factor alpha is critical in the modulation of vascular smooth muscle cell proliferation . Mol Med Rep. 2015; ; 11 : :4353.–4358. 32. Wang XH, Cai P, Wang MH, Wang Z. microRNA25 promotes osteosarcoma cell proliferation by targeting the cellcycle inhibitor p27 . Mol Med Rep. 2014; ; 10 : :855.–859. 33. Fossat N, Tourle K, Radziewic T, et al. C to U RNA editing mediated by APOBEC1 requires RNA-binding protein RBM47 . EMBO Rep. 2014; ; 15 : :903.–910. 34. Rayon-Estrada V, Harjanto D, Hamilton CE, et al. Epitranscriptomic profiling across cell types reveals associations between APOBEC1-mediated RNA editing, gene expression outcomes, and cellular function . Proc Natl Acad Sci USA. 2017; ; 114 : :13296.–13301. 35. Fossat N, Tam PP. Re-editing the paradigm of Cytidine (C) to Uridine (U) RNA editing . RNA Biol. 2014;; 11 : :1233.–7. 36. Severi F, Conticello SG. Flow-cytometric visualization of C>U mRNA editing reveals the dynamics of the process in live cells . RNA Biol. 2015; ; 12 : :389.–397. |