1.
Isaacs A, Lindenmann J. Virus interference. I. The interferon. Proc R Soc Lond B Biol Sci 1957; 147 : 258–67.
2.
Krause D, Silverman RH, Jacobsen H, et al. Regulation of ppp(A2’p)nA-dependent RNase levels during interferon treatment and cell differentiation. Eur J Biochem 1985; 146 : 611–8.
3.
Kerr IM, Brown RE, Ball LA. Increased sensitivity of cell-free protein synthesis to double-stranded RNA after interferon treatment. Nature 1974; 250 : 57–9.
4.
Brown GE, Lebleu B, Kawakita M, et al. Increased endonuclease activity in an extract from mouse Ehrlich ascites tumor cells which had been treated with a partially purified interferon preparation : dependence of double-stranded RNA. Biochem Biophys Res Commun 1976; 69 : 114–22.
5.
Clemens MJ, Williams BR. Inhibition of cell-free protein synthesis by pppA2’p5’A2’p5’A : a novel oligonucleotide synthesized by interferon-treated L cell extracts. Cell 1978; 13 : 565–72.
6.
Baglioni C, Minks MA, Maroney PA. Interferon action may be mediated by activation of a nuclease by pppA2’p5’A2’p5’A. Nature 1978; 273 : 684–7.
7.
Hovanessian AG, Brown RE, Kerr IM. Synthesis of low molecular weight inhibitor of protein synthesis with enzyme from interferon-treated cells. Nature 1977; 268 : 537–40.
8.
Hovanessian AG, Justesen J. The human 2’-5’oligoadenylate synthetase family : unique interferon-inducible enzymes catalyzing 2’-5’ instead of 3’-5’ phosphodiester bond formation. Biochimie 2007; 89 : 779–88.
9.
Zhou A, Hassel BA, Silverman RH. Expression cloning of 2-5A-dependent RNAase : a uniquely regulated mediator of interferon action. Cell 1993; 72 : 753–65.
10.
Tanaka N, Nakanishi M, Kusakabe Y, et al. Structural basis for recognition of 2’,5’-linked oligoadenylates by human ribonuclease L. EMBO J 2004; 23 : 3929–38.
11.
Cole JL, Carroll SS, Kuo LC. Stoichiometry of 2’,5’-oligoadenylate-induced dimerization of ribonuclease L. A sedimentation equilibrium study. J Biol Chem 1996; 271 : 3979–81.
12.
Nakanishi M, Goto Y, Kitade Y. 2-5A induces a conformational change in the ankyrin-repeat domain of RNase L. Proteins 2005; 60 : 131–8.
13.
Dong B, Silverman RH. A bipartite model of 2-5A-dependent RNase L. J Biol Chem 1997; 272 : 22236–42.
14.
Le Roy F, Salehzada T, Bisbal C, et al. A newly discovered function for RNase L in regulating translation termination. Nat Struct Mol Biol 2005; 12 : 505–12.
15.
Sidrauski C, Walter P. The transmembrane kinase Ire1p is a site-specific endonuclease that initiates mRNA splicing in the unfolded protein response. Cell 1997; 90 : 1031–9.
16.
Silverman RH. A scientific journey through the 2-5A/RNase L system. Cytokine Growth Factor Rev 2007; 18 : 381–8.
17.
Georgel P, Bahram S. Immunité innée antivirale : rôle des mécanismesToll-dépendants. Med Sci (Paris) 2006; 22 : 961–68.
18.
Delneste Y, Beauvillain C, Jeannin P. Immunité naturelle : structure et fonction des Toll-like receptors. Med Sci (Paris) 2007; 23 : 67–73.
19.
Malathi K, Dong B, Gale M Jr, Silverman RH. Small self-RNA generated by RNase L amplifies antiviral innate immunity. Nature 2007; 448 : 816–9.
20.
Bisbal C. Cliver les ARN du soi donne du punch à la réponse innée antivirale. Med Sci (Paris) 2008; 24 : 23–5.
21.
Liang SL, Quirk D, Zhou A. RNase L : its biological roles and regulation. IUBMB Life 2006; 58 : 508–14.
22.
Bisbal C, Silverman RH. Diverse functions of RNase L and implications in pathology. Biochimie 2007; 89 : 789–98.
23.
Silverman RH. Viral encounters with OAS and RNase L during the IFN antiviral response. J Virol 2007.
24.
Gribaudo G, Lembo D, Cavallo G, et al. Interferon action : binding of viral RNA to the 40-kilodalton 2’-5’- oligoadenylate synthetase in interferon-treated HeLa cells infected with encephalomyocarditis virus. J Virol 1991; 65 : 1748–57.
25.
Castelli JC, Hassel BA, Maran A, et al. The role of 2’-5’ oligoadenylate-activated ribonuclease L in apoptosis. Cell Death Differ 1998; 5 : 313–20.
26.
Diaz GM, Rivas C, Esteban M. Activation of the IFN-inducible enzyme RNase L causes apoptosis of animal cells. Virology 1997; 236 : 354–63.
27.
Zhou A, Paranjape J, Brown TL, et al. Interferon action and apoptosis are defective in mice devoid of 2’,5’- oligoadenylate-dependent RNase L. EMBO J 1997; 16 : 6355–63.
28.
Rusch L, Zhou A, Silverman RH. Caspase-dependent apoptosis by 2’,5’-oligoadenylate activation of RNase L is enhanced by IFN-beta. J Interferon Cytokine Res 2000; 20 : 1091–100.
29.
Le Roy F, Silhol M, Salehzada T, Bisbal C. Regulation of mitochondrial mRNA stability by RNase L is translation-dependent and controls IFNalpha-induced apoptosis. Cell Death Differ 2007; 14 : 1406–13.
30.
Li G, Xiang Y, Sabapathy K, Silverman RH. An apoptotic signaling pathway in the interferon antiviral response mediated by RNase L and c-Jun NH2-terminal kinase. J Biol Chem 2004; 279 : 1123–31.
31.
Chandrasekaran K, Mehrabian Z, Li XL, Hassel B. RNase-L regulates the stability of mitochondrial DNA-encoded mRNAs in mouse embryo fibroblasts. Biochem Biophys Res Commun 2004; 325 : 18–23.
32.
Bisbal C, Martinand C, Silhol M, et al. Cloning and characterization of a RNAse L inhibitor. A new component of the interferon-regulated 2-5A pathway. J Biol Chem 1995; 270 : 13308–17.
33.
Martinand C, Montavon C, Salehzada T, et al. RNase L inhibitor is induced during human immunodeficiency virus type 1 infection and down regulates the 2-5A/RNase L pathway in human T cells. J Virol 1999; 73 : 290–6.
34.
Camier S, Séraphin B. Détruisez ce message (ARN) après l’avoir lu ! Med Sci (Paris) 2007; 23 : 850–56.
35.
Li XL, Blackford JA, Judge CS, et al. RNase-L-dependent destabilization of interferon-induced mRNAs. A role for the 2-5A system in attenuation of the interferon response. J Biol Chem 2000; 275 : 8880–8.
36.
Khabar KS, Siddiqui YM, al-Zoghaibi F, et al. RNase L mediates transient control of the interferon response through modulation of the double-stranded RNA-dependent protein kinase PKR. J Biol Chem 2003; 278 : 20124–32.
37.
Bisbal C, Silhol M, Laubenthal H, et al. The 2’-5’ oligoadenylate/RNase L/RNase L inhibitor pathway regulates both MyoD mRNA stability and muscle cell differentiation. Mol Cell Biol 2000; 20 : 4959–69.
38.
Malathi K, Paranjape JM, Bulanova E, et al. A transcriptional signaling pathway in the IFN system mediated by 2’-5’-oligoadenylate activation of RNase L. Proc Natl Acad Sci USA 2005; 102 : 14533–8.
39.
Liu W, Liang SL, Liu H, et al. Tumour suppressor function of RNase L in a mouse model. Eur J Cancer 2007; 43 : 202–9.
40.
Carpten J, Nupponen N, Isaacs S, et al. Germline mutations in the ribonuclease L gene in families showing linkage with HPC1. Nat Genet 2002; 30 : 181–4.
41.
Urisman A, Molinaro RJ, Fischer N, et al. Identification of a novel Gammaretrovirus in prostate tumors of patients homozygous for R462Q RNASEL variant. PLoS Pathog 2006; 2 : e25.
42.
Bartsch DK, Fendrich V, Slater EP, et al. RNASEL germline variants are associated with pancreatic cancer. Int J Cancer 2005; 117 : 718–22.
43.
Kruger S, Silber AS, Engel C, et al. Arg462Gln sequence variation in the prostate-cancer-susceptibility gene RNASEL and age of onset of hereditary non-polyposis colorectal cancer : a case-control study. Lancet Oncol 2005; 6 : 566–72.
44.
Nijs J, Frémont M. Intracellular immune dysfunction in myalgic encephalomyelitis/chronic fatigue syndrome : state of the art and therapeutic implications. Expert Opin Ther Targets 2008; 12 : 281–89.
45.
Silberman B, Launay O. Prévention des infections à papillomavirus et du zona : nouveaux vaccins. Med Sci (Paris) 2007; 23 : 423–7.
46.
Thakur CS, Jha BK, Dong B, et al. Small-molecule activators of RNase L with broad-spectrum antiviral activity. Proc Natl Acad Sci USA 2007; 104 : 9585–90.
47.
Cussenot O, Cancel-Tassin G. Facteurs de risque génétiques pour le cancer de la prostate. Med Sci (Paris) 2004; 20 : 562–8.
48.
Darlix JL, Sitbon M. Le cancer de la prostate conduit à la découverte d’un nouveau rétrovirus infectieux humain. Med Sci (Paris) 2007; 23 : 690–1.



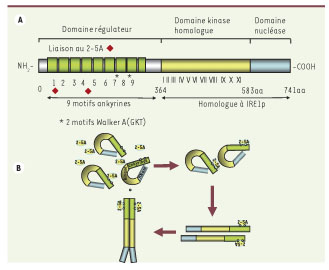
 ) et les 2 motifs Walker A (fixation de l’ATP ou du GTP) sont situés entre les motifs ankyrines R7 et R8. La moitié carboxy-terminale de la RNase L est homologue à la nucléase IRE1p. B. Modèle d’activation de la RNase L. La fixation du 2-5A induit un changement de conformation de l’extrémité amino-terminale de la protéine, ce qui permet un démasquage du domaine nucléase carboxy-terminal. Ce changement de conformation permet son homodimérisation et/ou son interaction avec d’autres protéines et son activation.
) et les 2 motifs Walker A (fixation de l’ATP ou du GTP) sont situés entre les motifs ankyrines R7 et R8. La moitié carboxy-terminale de la RNase L est homologue à la nucléase IRE1p. B. Modèle d’activation de la RNase L. La fixation du 2-5A induit un changement de conformation de l’extrémité amino-terminale de la protéine, ce qui permet un démasquage du domaine nucléase carboxy-terminal. Ce changement de conformation permet son homodimérisation et/ou son interaction avec d’autres protéines et son activation.